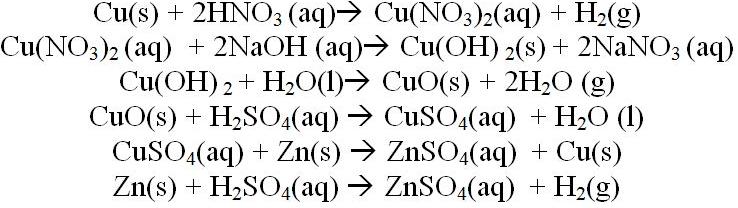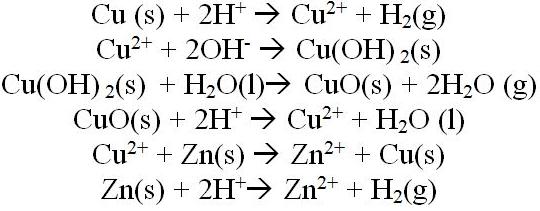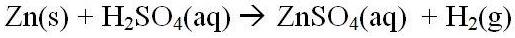Stoichiometry Using Copper
Purpose: The purpose is to see how the amount of copper (and copper itself) is altered after a series of reactions.
-Quantitative Data Table-
-Qualitative Data Table-
-Balanced Reactions- -In order of Use-
-Net Ionic Equations in Order of Above-
-Calculations-
Final Mass of Copper= Mass of Evaporating Dish and Copper - Mass of Evaporating Dish.
3.53 grams= 59.91 grams - 56.38 grams
Final Moles of Copper= 3.53 g Cu | 1 mol Cu | = .0555 mols Cu
| 63.55 g Cu |
Final Moles of Copper= 1.99 g Cu | 1 mol Cu | = .0313 mols Cu
| 63.55 g Cu |
Percent Yield of Copper = 3.53 g Cu x100 = 177 % yield
1.99 g Cu
3.53 grams= 59.91 grams - 56.38 grams
Final Moles of Copper= 3.53 g Cu | 1 mol Cu | = .0555 mols Cu
| 63.55 g Cu |
Final Moles of Copper= 1.99 g Cu | 1 mol Cu | = .0313 mols Cu
| 63.55 g Cu |
Percent Yield of Copper = 3.53 g Cu x100 = 177 % yield
1.99 g Cu
Conclusion
The conclusion reached in this experiment is that while it is the ideal situation that all of the copper would be conserved and no contaminants would be in it, this was not the case in the real situation. A major concern was originally that there would be copper wasted and the yield would be too low, while in reality there was contaminants left with the copper that increased the percent yield to above 100 percent. Stoichiometric calculations are useful in predictions, but in real life situations errors are going to be made in the experiment that distort the data received. In short, the percent yield was 177 percent, exceeding the real value of the copper and making it clear that some containments were left in the mix.
Discussion of Theory
The main theory used in the lab was the conservation of mass. While copper was subjected to many different types of reactions the mass of copper was projected to stay the same. Even when there were reactions in which copper was bonded to another substance, when it is produced as a solid copper in
the last reaction the mass of copper is supposed to have been the same.
This is the conservation of mass. However, as seen by the difference in mass at the beginning of the experiment and after, this was not the case in the real situation. The law of conservation was not broken; this was simply a culmination of errors that created a difference in mass. As seen by the sources of error section, there were many errors that could have occurred in this lab. The mass being greater than it was originally was signals that there were containments mixed in with the copper that were counted in the total mass at the end of the experiment. Sources of error included any extra ions that were not rinsed out of the mixture during the decanting process and copper being bonded to other elements.
The types of reactions used in the experiment were also used by the stoichiometry calculations vary incredibly. There were single and double replacement reactions as well as dehydration and redox reactions as well. These reactions allowed for many sources of error such as some things being limiting reactions and such, however it is assumed that if all reactions carried out completely and proper steps were taken the mass of copper would be the same mass before and after the reaction.
the last reaction the mass of copper is supposed to have been the same.
This is the conservation of mass. However, as seen by the difference in mass at the beginning of the experiment and after, this was not the case in the real situation. The law of conservation was not broken; this was simply a culmination of errors that created a difference in mass. As seen by the sources of error section, there were many errors that could have occurred in this lab. The mass being greater than it was originally was signals that there were containments mixed in with the copper that were counted in the total mass at the end of the experiment. Sources of error included any extra ions that were not rinsed out of the mixture during the decanting process and copper being bonded to other elements.
The types of reactions used in the experiment were also used by the stoichiometry calculations vary incredibly. There were single and double replacement reactions as well as dehydration and redox reactions as well. These reactions allowed for many sources of error such as some things being limiting reactions and such, however it is assumed that if all reactions carried out completely and proper steps were taken the mass of copper would be the same mass before and after the reaction.
Sources of Error
The final mass of copper in the experiment was too high compared to the initial amount of copper. Starting with 1.99 grams and ending with 3.53 was a major shock that led to many questions about the possible sources of error. The first being that the zinc was still reacting with the sulfuric acid when it was being decanted and that there was still leftover zinc left in the container. Another was that some water used for decanting could have had minerals in it that stuck in the container when it was receiving a steam bath. However many other sources of error led the group to believe that there would be less copper than originally used. These sources of error included spilling some of the copper out during the decanting process, copper clinging to the side of the beaker, and copper clinging to the stirring rod. It was unexpected that the yield would be too high, but considering that sources of error it makes sense that zinc left in the container would skew the results much more than a small spill would. The main assumption is that the bubbles in the beaker before the final decanting meant the reaction was still occurring, even after a 24 hour waiting period.
Questions
1. Why is the product of the reaction between copper and the nitric acid in step two placed in ice?
The main reason of placing ice on the mixture is to cool it, as stated in the directions. This is so when the mixture is mixed in with the addition of sodium hydroxide the reaction can occur normally (not too fast) and to make it easier to work with.
2. What type of reaction occured in steps 4, 7, and 9?
The reaction in step 4 is a double replacement reaction.
The reaction in step 7 is a double replacement reaction as well as a dehydration reaction (produces water).
The reaction in step 9 is a single replacement reaction as well as a redox reaction.
3. The reaction of excess zinc with sulfuric acid is a critical step in this investigation. Write a balanced equation for this reaction. What problems would arise from an incomplete reaction?
Problems that could arise from an incomplete reaction include leftover amounts of zinc which would mix in with the copper during the weighing, or excess amounts of the sulfuric acid which would simply be decanted out. The main issue would be the leftover zinc, as that would be confused with copper when measured and would lead to a high percent yield. This was assumed to be seen in the experiment performed.
4. What ions did you removed when you washed the CuO?
In this stage the ions removed were any excess hydroxide ions and perhaps some uncreacted copper ions as well that were not included in the reaction. Water was removed as well as part of the washing process.
5. What form of copper is present in the beaker after you added the Sulfuric Acid?
After the first addition of sulfuric acid the copper was in the form of copper sulfate, which is an aqueous solution.
6. What ions did you remove when you washed the precipitated copper?
The ions removed in this step were zinc and sulfate ions as part of the zinc sulfate mixture that was "aq" as well as copper and sulfate ions that may be part of the unreacted part of the last reaction which included copper sulfate.
The main reason of placing ice on the mixture is to cool it, as stated in the directions. This is so when the mixture is mixed in with the addition of sodium hydroxide the reaction can occur normally (not too fast) and to make it easier to work with.
2. What type of reaction occured in steps 4, 7, and 9?
The reaction in step 4 is a double replacement reaction.
The reaction in step 7 is a double replacement reaction as well as a dehydration reaction (produces water).
The reaction in step 9 is a single replacement reaction as well as a redox reaction.
3. The reaction of excess zinc with sulfuric acid is a critical step in this investigation. Write a balanced equation for this reaction. What problems would arise from an incomplete reaction?
Problems that could arise from an incomplete reaction include leftover amounts of zinc which would mix in with the copper during the weighing, or excess amounts of the sulfuric acid which would simply be decanted out. The main issue would be the leftover zinc, as that would be confused with copper when measured and would lead to a high percent yield. This was assumed to be seen in the experiment performed.
4. What ions did you removed when you washed the CuO?
In this stage the ions removed were any excess hydroxide ions and perhaps some uncreacted copper ions as well that were not included in the reaction. Water was removed as well as part of the washing process.
5. What form of copper is present in the beaker after you added the Sulfuric Acid?
After the first addition of sulfuric acid the copper was in the form of copper sulfate, which is an aqueous solution.
6. What ions did you remove when you washed the precipitated copper?
The ions removed in this step were zinc and sulfate ions as part of the zinc sulfate mixture that was "aq" as well as copper and sulfate ions that may be part of the unreacted part of the last reaction which included copper sulfate.