Conclusion
The conclusion of this lab is that chemical reactions can be observed by the human eye, but not always with the most accuracy. While some reactions were labeled as DNR and others were labeled as precipitate, color change, or gas there was no accurate way to measure the reaction other than the human eye. When discerning between an air bubble and gas created by reaction, error was certainly a large factor.
The conclusion of this lab is that chemical reactions can be observed by the human eye, but not always with the most accuracy. While some reactions were labeled as DNR and others were labeled as precipitate, color change, or gas there was no accurate way to measure the reaction other than the human eye. When discerning between an air bubble and gas created by reaction, error was certainly a large factor.
Discussion of theory
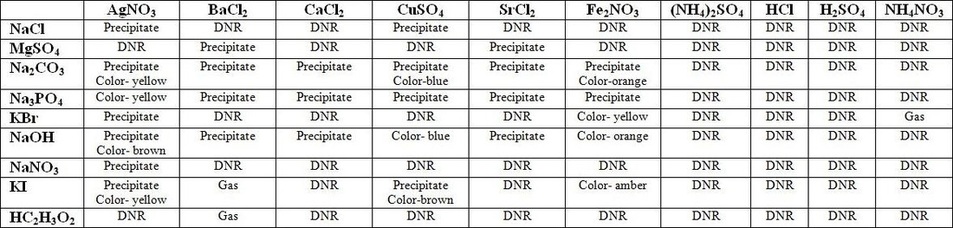
The method used in this lab begins with identifying which reactions happened with the human eye, essentially any physical changes that occur accompanying a reaction. This means identifying any precipitate or gasformation along with any changes in color. These cover two of the metathesis reactions, with the exception being the formation of water. The purpose of this is to find an associated pattern of what ions react and form a precipitate or gas (the pattern or generalizations of solubility are found in the analysis questions, question 1). Using the data recorded, it is possible to gauge the solubility of a cation or anion. Any rows or columns of precipitate formation, gas formation or color change signify a higher change of the ion being insoluble, whereas a row or column of “DNR” signifies a likelihood of an ion forming a soluble compound. Ideally these reactions will serve as evidence for the solubility rules, hopefully following and in a sense proving all of the rules provided.
The method used in this lab begins with identifying which reactions happened with the human eye, essentially any physical changes that occur accompanying a reaction. This means identifying any precipitate or gasformation along with any changes in color. These cover two of the metathesis reactions, with the exception being the formation of water. The purpose of this is to find an associated pattern of what ions react and form a precipitate or gas (the pattern or generalizations of solubility are found in the analysis questions, question 1). Using the data recorded, it is possible to gauge the solubility of a cation or anion. Any rows or columns of precipitate formation, gas formation or color change signify a higher change of the ion being insoluble, whereas a row or column of “DNR” signifies a likelihood of an ion forming a soluble compound. Ideally these reactions will serve as evidence for the solubility rules, hopefully following and in a sense proving all of the rules provided.
Sources of Error
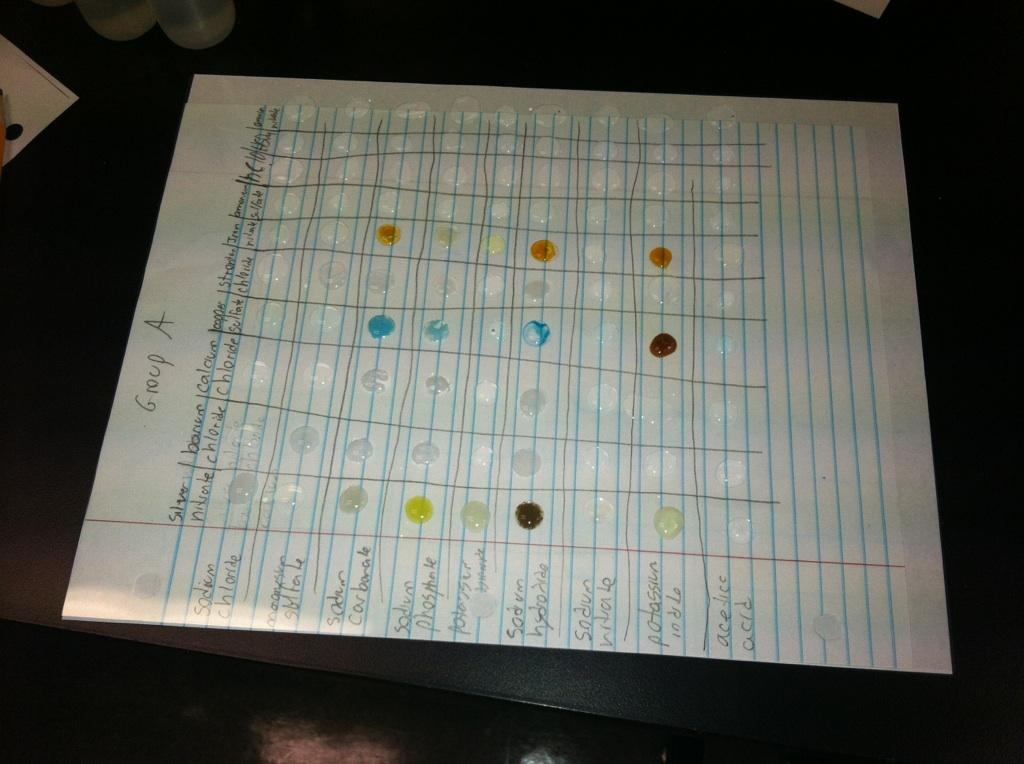
Several sources of error are to be accounted for within this lab. The first was the accuracy of the human eye in discerning reactions, as stated in the conclusion. It was extremely difficult to tell the difference between gas and a simple air bubble, just as it was to tell if color change happened jointly with the formation of a precipitate. There was little error when it came to mixing the chemicals, as the drops were usually contained to their squares, however as seen by the photo above there was a drop that melded with the one above it. This may have been the source of error when deciding if those chemicals reacted. Also there may have been some error with the mole to mole ratio, such as an extremely low limiting that reactor that limited the reaction so severely that it was not inheritably clear that a reaction was taking place.
Several sources of error are to be accounted for within this lab. The first was the accuracy of the human eye in discerning reactions, as stated in the conclusion. It was extremely difficult to tell the difference between gas and a simple air bubble, just as it was to tell if color change happened jointly with the formation of a precipitate. There was little error when it came to mixing the chemicals, as the drops were usually contained to their squares, however as seen by the photo above there was a drop that melded with the one above it. This may have been the source of error when deciding if those chemicals reacted. Also there may have been some error with the mole to mole ratio, such as an extremely low limiting that reactor that limited the reaction so severely that it was not inheritably clear that a reaction was taking place.
Analysis Questions
1. Using your data, write a list of general statements describing the solubility of specific cations and anions.
Cations that do not seem to usually form soluble chemicals are Silver, Lead, and Mercury. Anions that usually are not soluble are Phosphate, Carbonate, Sulfide, and Hydroxide. As seen in the lab data, with most cases these anions and cations formed insoluble compounds, the precipitate. Group 1 cations usually were soluble along with ammonium.
1. Using your data, write a list of general statements describing the solubility of specific cations and anions.
Cations that do not seem to usually form soluble chemicals are Silver, Lead, and Mercury. Anions that usually are not soluble are Phosphate, Carbonate, Sulfide, and Hydroxide. As seen in the lab data, with most cases these anions and cations formed insoluble compounds, the precipitate. Group 1 cations usually were soluble along with ammonium.
3. Give a brief description of the three types of metathesis reactions and give at least one example from this lab.
One of the three types of metathesis reactions is precipitation, where a product forms that is insoluble. Another type is neutralization where a product formed is a neutral water molecule, and the last is a gas formation reaction that forms a gas as a product. In this lab, reactions 1-10 in question 2 are all precipitation reactions. This is because in all of the reactions there was a solid precipitate being formed as a product from two aqueous solutions.
4. Why are some things soluble in water, while others are not?
The simple answer to this question is the phrase “like dissolves like”, which is used to express that polar molecules dissolve other polar molecules and non-polar molecules dissolve other non-polar molecules. Water is a polar molecule meaning that it can dissolve other polar molecules. It can also dissolve ionic bonds with the processes of dissociation and hydration. Essentially the bonds of an ionic compound are broken and hydrogen surrounds the negative ions and the oxygen
surrounds the positive ions in hydration.
5. Explain why a double replacement reaction that produces two aqueous products is considered a "no reaction".
This is considered a "no reaction" because when two aqueous solutions create a new set of aqueous solutions it is, essentially, the same mixture of ions floating around in the solution. See diagram for a visual description, as describing in words is often times inadequate. The white is liquid, while pink is precipitate.
One of the three types of metathesis reactions is precipitation, where a product forms that is insoluble. Another type is neutralization where a product formed is a neutral water molecule, and the last is a gas formation reaction that forms a gas as a product. In this lab, reactions 1-10 in question 2 are all precipitation reactions. This is because in all of the reactions there was a solid precipitate being formed as a product from two aqueous solutions.
4. Why are some things soluble in water, while others are not?
The simple answer to this question is the phrase “like dissolves like”, which is used to express that polar molecules dissolve other polar molecules and non-polar molecules dissolve other non-polar molecules. Water is a polar molecule meaning that it can dissolve other polar molecules. It can also dissolve ionic bonds with the processes of dissociation and hydration. Essentially the bonds of an ionic compound are broken and hydrogen surrounds the negative ions and the oxygen
surrounds the positive ions in hydration.
5. Explain why a double replacement reaction that produces two aqueous products is considered a "no reaction".
This is considered a "no reaction" because when two aqueous solutions create a new set of aqueous solutions it is, essentially, the same mixture of ions floating around in the solution. See diagram for a visual description, as describing in words is often times inadequate. The white is liquid, while pink is precipitate.