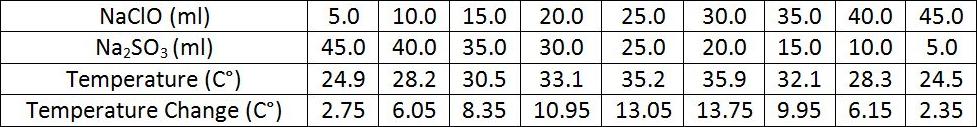
Finding the Ratio of Moles of Reactants in a Chemical Reaction
Purpose: The purpose of this expirement was to find the ratio of moles of reactants in a chemical reaction by the method of continuous variations.
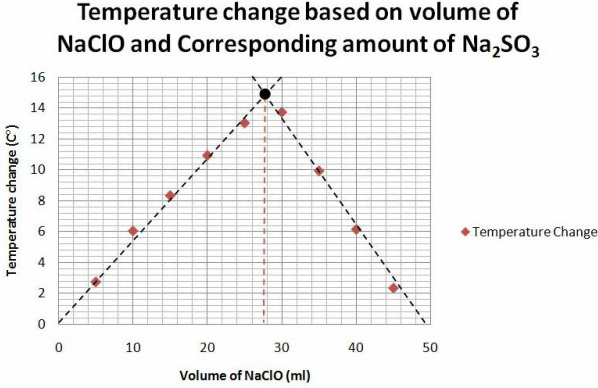
The graph's x-axis has the volume of the NaClO on it. For the corresponding amount of Sodium Sulfite it is necessary to subtract the NaClO ml by 50.
Analysis Questions
1. Why did you have to keep a constant volume of the reactants?
In order to accurately gauge the amount of temperature change from the reaction, the reaction reactants total volume must be of the same size. If the total amount of reactants was different in different trials the temperature change would no longer be comparable.
2. What is meant by the term "Limiting Reagent"?
The limiting reagent is the substance that was completely consumed by the reaction. When this happens the reaction can no longer continue no matter what amount of the other substance is left, thus limiting the reaction.
3. Which measurement limits the precesion of your data: temperature or volume? Explain.
The volume limits the precision because while we did control the volume, during the pouring of the liquids it could have been off. With these amounts being off the lines may be slightly skewed causing us to receive the wrong mole to mole ratio. Also the temperature may have limited the precision because we may have looked at the thermometer incorrectly or we may have let it sit too long or not for long enough. In essence both of these limit the precision of the data.
4. Which reactant is the limiting reagent along the upward sloping line of your graph? Which is the limiting reagent along the downward sloping line?
The NaClO is the limiting reagent along the upward slope because as we add more it reacts more, meaning that it was limiting the other reactions. Along the downward slope the limiting reagent was the Sodium Sulfite because even though we had enough of NaClO to react it didn't because there was not enough of the other reactant.
5. What phsyical properties, other than temperature change, could use the method of continuous variations?
Other physical properties that can be observed are the mass of the precipitate that forms, the intensity of color change, or the volume of gas that evolved.
6. Why is it more acurate to use the point of the two lines to find the mole to mole ratio, rather than the ratio associated with the greatest temperature change?
It is more accurate to use the point of the two lines instead of just the highest temperature change because we did not test all value combinations of the two reactants,we tested just enough to get the accurate lines of best fit and connect them. It would not be appropriate because there are mole to mole ratios between the ratios that we used.
1. Why did you have to keep a constant volume of the reactants?
In order to accurately gauge the amount of temperature change from the reaction, the reaction reactants total volume must be of the same size. If the total amount of reactants was different in different trials the temperature change would no longer be comparable.
2. What is meant by the term "Limiting Reagent"?
The limiting reagent is the substance that was completely consumed by the reaction. When this happens the reaction can no longer continue no matter what amount of the other substance is left, thus limiting the reaction.
3. Which measurement limits the precesion of your data: temperature or volume? Explain.
The volume limits the precision because while we did control the volume, during the pouring of the liquids it could have been off. With these amounts being off the lines may be slightly skewed causing us to receive the wrong mole to mole ratio. Also the temperature may have limited the precision because we may have looked at the thermometer incorrectly or we may have let it sit too long or not for long enough. In essence both of these limit the precision of the data.
4. Which reactant is the limiting reagent along the upward sloping line of your graph? Which is the limiting reagent along the downward sloping line?
The NaClO is the limiting reagent along the upward slope because as we add more it reacts more, meaning that it was limiting the other reactions. Along the downward slope the limiting reagent was the Sodium Sulfite because even though we had enough of NaClO to react it didn't because there was not enough of the other reactant.
5. What phsyical properties, other than temperature change, could use the method of continuous variations?
Other physical properties that can be observed are the mass of the precipitate that forms, the intensity of color change, or the volume of gas that evolved.
6. Why is it more acurate to use the point of the two lines to find the mole to mole ratio, rather than the ratio associated with the greatest temperature change?
It is more accurate to use the point of the two lines instead of just the highest temperature change because we did not test all value combinations of the two reactants,we tested just enough to get the accurate lines of best fit and connect them. It would not be appropriate because there are mole to mole ratios between the ratios that we used.
Conclusion
We found that the ideal amount of NaClO for this reaction was extremely close to 28 ml. Subtracting this from 50 because we kept the total volume constant for every trial that means that the ideal amount of Sodium Sulfite was 22 ml. Using these amounts gathered we continued to find the mole to mole ratio by dividing 28 ml by 22 ml to get the Sodium Sulfite to a subscript of 1 and the NaClO to 1.3. Multiplying both the 3 to get whole numbers, as is prefered, the ratio came out to be 4:3 for NaClO and Sodium Sulfite respectably.
Discussion of Theory
This method of continuous variations allowed us to get the mole to mole ratio by experimenting with different mole to mole combinations. While the mole to mole ratio can not be accurately guaged by just trying different mole ratios at different volumes we kept the total volume of the mixture the same. With this step taken care of, by experimenting with different volumes we managed to gain enough data to construct lines of best fit. The data gathered was the temperature change, which shows the intensity of the reaction. With the lines of best fit we constucted we could connect them and use the intersection as the most accurate point to calculate the mole to mole ratio. This point is where the temperature change was the greatest and that signifies that both of the reactants we used nearly completely.